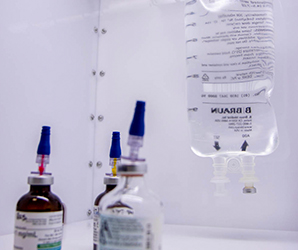
IV Hood
IV Hoods provide a positive-pressure enclosure to protect the product and users from cross-contamination of airborne particulate. IV Hoods supply reduced particulate air to the workspace, creating an ISO Class 5 cleanroom environment suitable for IV Bag preparation. Healthcare professionals should prepare IV bags in a sterile environment to produce the highest quality product and safeguard patients’ health and safety.
Laminar flow cabinets create a clean work enclosure for IV bag preparation through a positive pressure or horizontal laminar flow pattern. A horizontal laminar flow pattern forms when the system draws in ambient air through the HEPA filter and supplies clean air to the IV Hood. Horizontal laminar airflow means the air moves in the same direction at a constant velocity to help minimize disruptions to the compounding process. The IV Hood features a stainless steel IV Bag rod for convenient and sterile temporary storage. Rods are available with 7 or 10 hooks, depending on the size of the hood. Optional carts are also available to improve the portability of the system.
USP 797 IV Hood
Model # SS-XXX-XXX

Unit Configurations
Additional Configurations
IV Preparation
IV preparation entails compounding medication, vitamins, and supplements into an intravenous fluid to be supplied directly to the bloodstream. IV or infusion therapy has grown in popularity for outpatient services and offers immediate therapeutic responses to treat a variety of medical conditions.
For advance spiking of IV bags, facilities must use a USP 797-certified solution that provides ISO Class 5 cleanroom air in a unidirectional airflow. For immediate use, portable clean rooms or reduced particulate workspaces with ISO Class 5 air can help prevent IV bag contamination. Following aseptic techniques and using proper engineering controls for IV bag prep helps preserve medication quality and protect patients’ safety.
| Particle Size | Maximum # of Particles |
| ≥ 0.1 µm | 100,000 |
| ≥ 0.2 µm | 23,700 |
| ≥ 0.3 µm | 10,200 |
| ≥ 0.5 µm | 3,520 |
IV Hoods, or Laminar Flow Clean Benches, provide an ISO Class 5 environment. In order to meet ISO Class 5 standards, the filtration system must meet strict limits on the maximum number of particles sized 0.1 to 0.5 microns per cubic feet in the air. The number of particles must be limited to ensure a clean and hygienic work surface for IV bag infusion or preparation. The ISO standard limits on particles for an ISO Class 5 cleanroom are detailed in the table.
Laminar Flow Hood Cleaning
Proper cleaning procedures of the IV hood can help preserve medication quality. Procedures include cleaning, disinfecting, and applying a sporicidal agent to the hood as well as nearby surfaces. Operators should follow the outlined steps below to properly prevent contamination to the IV Hood.
- Clean – First, wipe down the hood with sterile water and low-lint wipers. Then, in order to remove residue, dirt, and debris, operators must use a cleaning agent on all surfaces of the interior of the hood.
- Disinfect – Remove fungi, viruses, and bacteria with a sterile disinfectant such as sterile 70% IPA.
- Dry – Allow surfaces to air dry fully before beginning compounding procedures.
All surfaces in the IV Hood must be cleaned and disinfected at the beginning and end of shifts as well as when contamination is suspected. However, the horizontal work surface in an IV hood must be cleaned and disinfected every 30 minutes or in between compounding activities, if one compounding activity takes longer than 30 minutes.
Other surfaces near the IV Hood should be cleaned on a schedule to prevent contamination. Sinks, pass-through chambers, areas outside of the hood, and floors need to be cleaned and disinfected daily. However, the walls, doors, door frames, ceilings, storage bins, and shelving only need to be cleaned and disinfected monthly.
For all surfaces, including inside the IV Hood, a sporicidal agent must be applied on a monthly basis to remove bacterial and fungal spores.
Surface Sampling
Monthly surface sampling helps check for proper cleaning and disinfecting methods. Sampling is taken after compounding but before cleaning the areas. Areas to sample include interior of the hood, area near the hood, equipment in the hood, frequently touched areas, and pass-through chambers. After sampling, areas need to be cleaned and disinfected.
12in. Portable Clean Room
Product Video
Hood Dimensions:
Outer: 22.25” x 19.5” x
21.75”
Interior: 15.5” x 17” x 17”
Access Area: 17” x 17”
Hood Material:
16-gauge Stainless Steel
Total Weight:
Approx. 74 lbs.
Light Intensity:
1800 lux at the work surface
Electrical:
115/1/60, 2.5 amps
Sound Level:
61 dba @ 3 ft.
Max Operating Temperature:
Not to exceed 170 degrees Fahrenheit
Warranty:
Limited one-year warranty from date of shipment on defects due to materials or workmanship.
Product Video
Hood Dimensions:
- O.D: 19"D x 20" W x 24"H
- 18"D x 18"W x 15.5"H
Hood Material:
- Polyethylene Terephthalate Glycol (PETG)
- Base: 0.250" Clear Polycarbonate
Blower Dimensions:
7.5" L x 7.5" W x 8.5" H
Total Weight:
Approx. 11 lbs.
Motor:
Fractional Horsepower
Electrical:
115/1/60, .3 amps
220/1/50, .2 amps
Sound Level:
Approx. 55 dba @ 3' from hood inlet
Max Operating Temperature:
Not to exceed 170 degrees Fahrenheit
Warranty:
Limited two-year warranty from date of shipment on defects due to materials or workmanship.
Patented Design:
U.S. PATENT #5,843,197
Product Video
Hood Dimensions:
- O.D.: 18.5"D x 20" W x 19"H
- I.D.: 18"D x 18"W x 18"H
Hood Material:
- Sides: .250" White Acrylic
- Base: .500" White High Density Polyethylene
- Top: .500" Clear Polycarbonate
Front Windows:
0.250" Clear Polycarbonate
Blower Dimensions:
10.5" L x 8" W x 8.5" H
Total Weight:
Approx. 35 lbs.
Motor:
Fractional Horsepower
Electrical:
115/1/60, .46 amps
Sound Level:
64 dba @ 3ft. from hood inlet
Max Operating Temperature:
Not to exceed 170 degrees Fahrenheit
Warranty:
6 Month Warranty on
Fan/Motor. 24 Month
Warranty on other parts.
Patented Design:
U.S. PATENT #5,843,197
Product Video
Hood Dimensions:
- O.D: 24"D x 26"W x 25"H
- I.D.: 23.5"D x 24"W x 24"H
Hood Material:
- Sides: .250" White Acrylic
- Base: .500" White High Density Polyethylene
- Top: .500" Clear Polycarbonate
Front Windows:
0.250" Clear Polycarbonate
Blower Dimensions:
13" L x 13" W x 14.5" H
Total Weight:
Approx. 95 lbs.
Motor:
Fractional Horsepower
Electrical:
115/1/60, 2 amps
220/1/50, .75 amps
Sound Level:
55 dba @ 3ft. from hood inlet
Max Operating Temperature:
Not to exceed 170 degrees Fahrenheit
Warranty:
Limited two-year warranty from date of shipment on defects due to materials or workmanship.
Patented Design:
U.S. PATENT #5,843,197
Product Video
Hood Dimensions:
- O.D: 24"D x 32"W x 25"H
- I.D.: 23.5"D x 30"W x 24"H
Hood Material:
- Sides: .250" White Acrylic
- Base: .500" White High Density Polyethylene
- Top: .500" Clear Polycarbonate
Front Windows:
0.250" Clear Polycarbonate
Blower Dimensions:
13" L x 13" W x 14.5" H
Total Weight:
Approx. 100 lbs.
Motor:
Fractional Horsepower
Electrical:
115/1/60, 2 amps
220/1/50, .75 amps
Sound Level:
Approx. 55 dba @ 3' from hood inlet
Max Operating Temperature:
Not to exceed 170 degrees Fahrenheit
Warranty:
Limited two-year warranty from date of shipment on defects due to materials or workmanship.
Patented Design:
U.S. PATENT #5,843,197
Product Video
Hood Dimensions:
- O.D: 24"D x 42W" x 25"H
- I.D.: 23.5"D x 40"W x 24"H
Hood Material:
- Sides: .250" White Acrylic
- Base: .500" White High Density Polyethylene
- Top: .500" Clear Polycarbonate
Front Windows:
0.250" Clear Polycarbonate
Blower Dimensions:
13" L x 13" W x 14.5" H
Total Weight:
Approx. 115 lbs.
Motor:
Fractional Horsepower
Electrical:
115/1/60, 2 amps
220/1/50, .75 amps
Sound Level:
Approx. 55 dba @ 3' from hood inlet
Max Operating Temperature:
Not to exceed 170 degrees Fahrenheit
Warranty:
Limited two-year warranty from date of shipment on defects due to materials or workmanship.
Patented Design:
U.S. PATENT #5,843,197
Quick Compare
|
Generation I |
Generation II |
Variable Speed Controller Standard |
No |
Yes |
Easy Filter Change |
Yes |
Yes |
Running Indicator Light |
No |
Yes |
Airflow |
Up to 350 CFM (HEPA) |
50 – 400 CFM (HEPA) |
Prefilter |
Yes – MERV 8 |
No |
Main Filters |
HEPA or ULPA |
HEPA or ULPA |
Electrical |
115/1/60, 2 amps |
115/1/60. 1.65 amps |
Sound Level |
55 dba |
50 - 65 dba |
ISO Classification |
Up to ISO Class 5 |
Up to ISO Class 5 |


Portable Clean Room with UVC Controls
SAS Portable Clean Rooms with UVC utilize the documented power of UV lights to actively disenfect clean room environments from pathogens, germs, viruses and other hazardous bacteria. Offering a simple user interface, the Portable Clean Room with UVC is essential in protecting workers from the environment they are working with, and the respirable hazards they may be exposed to.
Accessories
Resources
- Clean Room Hoods Applications
- Testimonial: Research in the Peruvian Rainforest
- Testimonial: Stem Cell Therapy
- Testimonial: Assembly of Projection Lenses in Portable Clean Rooms
- Testimonial: Vertical Laminar Flow Hood for Fungal Cell Culture
GSA
- GSA Contract #: GS-07F-0039X Category 66: SCIENTIFIC EQUIPMENT and SERVICES
- SIN 66-147: Laboratory Fume Hoods, Accessories & Options
- You will find the GSA logo on eligible product pages within our website. To obtain a Quote or receive additional information on these products, either:
- Fill out our feedback form.
- Visit our company profile in the GSA ELibrary and click on “View Items Available” for an itemized list of available products at GSA Advantage!


Q&A
What is laminar air flow?
Laminar air flow is when a stream of constant air is provided with the same speed and direction, with no crossover of air stream. The Vertical Laminar Flow Hood is rated as an ISO Class 5 clean room cabinet. It protects the process by utilizing a vertical laminar flow pattern and dual-stage filtration system that delivers positive-pressure, purified air through a pre-filter and main filter. The main filter is typically either a HEPA or ULPA filter depending on the efficiency desired. The HEPA filter is up to 99.97% efficient on particles 0.3 microns and larger, while the ULPA filter is up to 99.9995% efficient on particles 0.12 microns and larger.

Request A Quote or Contact Us
If you prefer, you may contact us by calling 800-799-4609, or e-mail us at sales@sentryair.com.
Name and Email are required.

 Made in the USA
Made in the USA